|
The hydrogen ion linked with acid characteristics of some compounds in water solution is better accurately depicted using the hydronium ion. The electron pair geometry is shown as tetrahedral, and the molecular geometry is shown as a trigonal pyramid. The third hydrogen atom connects to the molecule of water as a hydrogen ion (with no electrons) to the oxygen’s lone pair. The water molecule, on the other hand, has two hydrogen atoms and two lone electron pairs. The Lewis diagram in this example, H3O+, shows O at the centre with one lone electron pair with three hydrogen atoms attached. The lone electron pairs cause a modest compression to a 107o bond angle by exerting more repulsion on the three bonding hydrogen atoms.īecause the lone electron pair is invisible while looking at molecular geometry, the molecule has trigonal pyramidal molecular geometry.Because the boron hydride molecule lacked a lone electron pair, it had a flat trigonal planar molecular geometry. This is the geometry of a tetrahedral electron pair. At a roughly 109o bond angle, the three hydrogen atoms as well as the lone electron pair are as far apart as feasible. 
This leaves a single electron pair unattached to any other atom. 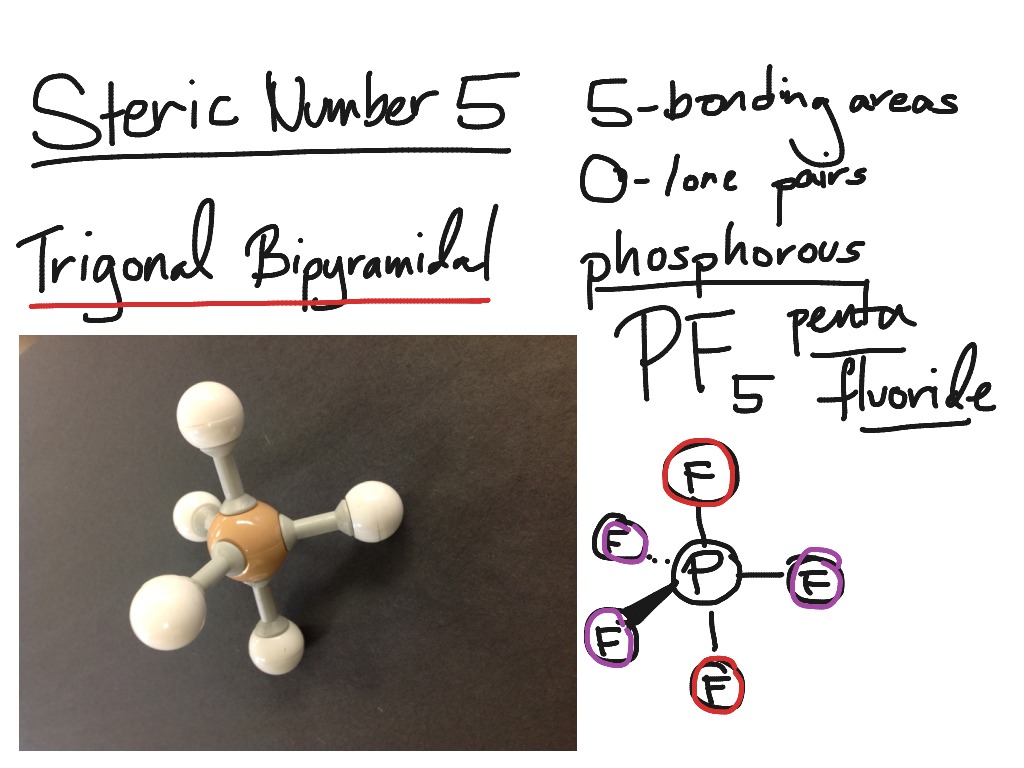
Because nitrogen has 5 valence electrons, it requires 3 more electrons from three hydrogen atoms to fulfil its octet. The molecular geometry of NH3 is an instance of trigonal pyramidal molecular geometry that derives from tetrahedral electron pair geometry. Phosphorus pentafluoride (PF5) or phosphorus pentachloride (PCl5) inside the gas phase are two examples of this molecular geometry. 
Because there is no geometrical configuration with five terminal atoms in equivalent places, the bond angles surrounding the central atom are not identical in this geometry (see also pentagonal bipyramid). A trigonal bipyramid creation is a molecular geometry in chemistry that has one atom in the centre and five additional atoms in the corners of a triangular bipyramid.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
